Chemical EquilibriumHard
Question
In the system, LaCl3(s) + H2O(g) + heat ⇋ LaClO(s) + 2HCl(g), equilibrium is established. More water vapour is added to restablish the equlibrium. The pressure of water vapour is doubled. The factor by which pressure of HCl is changed is:
Options
A.2
B.√2
C.√3
D.√5
Solution
LaCl3(s) + H2O(g) + heat → LaClO(s) + 2HCl(g)
Kp =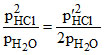 p′HCl = √2 PHCl.
p′HCl = √2 PHCl.
Kp =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
What will be the enthalpy change for the reaction (in kcal/mol)2H2(g) + O2(g) + 2H2O(l) → 4H+(aq) + 4OH- (aq.)Give...Iron fillings and water were placed in a 5 L vessel and sealed. The tank was heated to 1000oC. Upon analysis, the tank w...Which of the following statements is correct for a reversible process in a state of equilibrium ?...The reaction A(g) + B(g) $\rightleftharpoons$C(g) + D(g) occurs in a single step. The rate constant of forward reaction ...The degree of dissociation of PCl5 (a) obeying the equilibrium, PCl5 ⇋ PCl3 + Cl2, is approximately related to the...