Heat and Thermal ExpansionHard
Question
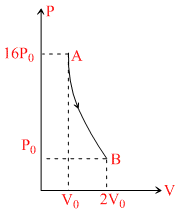
Figure demonstrates a poly tropic process (i.e. PVn = constant) for an ideal gas. The work done by the gas will be in process AB is

Options
A.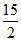 P0V0
P0V0
B.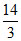 P0V0
P0V0
C.8 P0V0
D.insufficient information
Solution
For a poly tropic process PVn = constant ⇒ 16 P0V0n = P0(2V0)n ⇒ n = 4
Work done =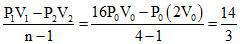
Work done =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Four moles of hydrogen , teo moles of helium and one mole of water vapor from an ideal gas mixture. What is the molar sp...In a mercy = glass thermometer the cross - section of the capillary portion is A0 and the volume of the bulb is V0 at 27...Given T - P curve for three processes. If initial and final pressure are same for all processes than work done in in pro...n moles of an ideal triatomic linear gas undergoes a proses in which the temperature changes with volume as T = k1V2 whe...Figure shows the adiabatic curve on log-log scale performed on a ideal gas. The gas must be :-...