Heat and Thermal ExpansionHard
Question
Figure shows the adiabatic curve on log-log scale performed on a ideal gas. The gas must be :-
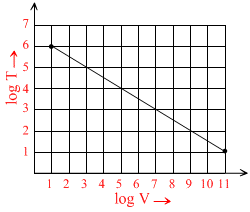

Options
A.Monoatomic
B.Diatomic
C.A mixture of monoatomic and diatomic
D.A mixture of diatomic and polyatomic
Solution
For adiabatic process TVγ-1 = constant
log T + (γ + 1) log V = constant ⇒ slope = - (γ + 1) = -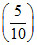 ⇒ γ =
⇒ γ = 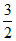
For monoatomic gas γ =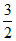 γ =
γ = 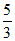 ; For diatomic gas γ =
; For diatomic gas γ = 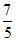 As
As 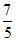 < γ =
< γ = 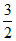 <
< 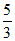
Hence, the gas must be a mixture of monoatomic & diatomic gas.
log T + (γ + 1) log V = constant ⇒ slope = - (γ + 1) = -
For monoatomic gas γ =
Hence, the gas must be a mixture of monoatomic & diatomic gas.
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A vessel is partitioned two equal halves by a fixed diathermy separator. Two different ideal gasses are filled in left (...A thermocouple of negligible resistance produces an e.m.f of 40 μ V/oC in the linear range of temperature. A galvan...The reversible expansion of an ideal gas under adiabatic and isothermal conditions is shown in the figure. Which of the ...Two long, thin solid cylinders are evidential in size, but they are of different substances with two different thermal c...A point source of heat of power P is placed at the center of a spherical shell of mean radius R the material of the shel...