Chemical EquilibriumHard
Question
For given two equilibrium attained in a container which are correct if degree of dissociation of A and A′ are α and α′.
A(s) ⇋ 2B(g) + C(g); Kp1 = 8 × 10-2
A′(s) ⇋ 2B + D(g); Kp2 = 2 × 10-2
A(s) ⇋ 2B(g) + C(g); Kp1 = 8 × 10-2
A′(s) ⇋ 2B + D(g); Kp2 = 2 × 10-2
Options
A.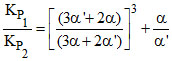
B.P′C/P′D = 4
C.P′B = 2P′C + 2P′D
D.α > α′
Solution
A(s) 2B(g) + C(s)
2α + 2α′ α
Aα(s) 2B + D
2α + 2α α′
Kp1 = (2α + 2α′) × α ×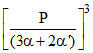
Kp2 = (2α + 2α′) ×α′ ×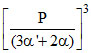
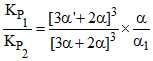
Also, KP1 = (P′B)2 × P′C
KP2 = (P′B)2 × P′p
or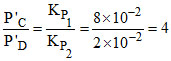
Also, P′B = 2 × (P′C + P′D)
KP1 > KP2 ∴ α > α1
2α + 2α′ α
Aα(s) 2B + D
2α + 2α α′
Kp1 = (2α + 2α′) × α ×
Kp2 = (2α + 2α′) ×α′ ×
Also, KP1 = (P′B)2 × P′C
KP2 = (P′B)2 × P′p
or
Also, P′B = 2 × (P′C + P′D)
KP1 > KP2 ∴ α > α1
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
When calcium acetate is dissolved in water, heat is evolved. If the temperature is raised, then the solubility of calciu...Carbon monoxide in water gas reacts with steam according to the following reaction.CO(g) + H2O(g) $\rightleftharpoons$CO...For the reaction I2(g) $\rightleftharpoons$2I(g), KC = 1.0 × 10−2 mol lit−1. What volume of the vessel should be taken s...Consider the following gaseous equilibrium in a closed container of volume "V" at T(K).$$P_{2}(\text{ }g) + Q_{2}(\text{...At constant temperature, the equilibrium constant (Kp) for the decomposition reaction N2O4 $\rightleftharpoons$ 2NO2 is...