Heat and Thermal ExpansionHard
Question
Figure shows the variation of the internal energy U with density ρ of one mole of an ideal monatomic gas for thermodynamic cycle ABCA. Here process AB is a a part of rectangular hyperbola :-
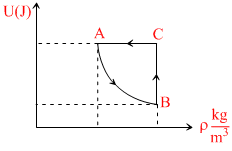

Options
A.process AB is isothermal & net work in cycle is done by gas.
B.process AB is isoberic & net work in cycle is done by gas.
C.process AB is isoberic & net work in cycle is done on the gas.
D.process AB is adiabatic & net work in cycle is done by gas.
Solution
For the process AB : = constant (hyperbola)
U =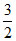 RT (monoatomic ideal gas);
RT (monoatomic ideal gas); 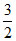 ρ RT = constant
ρ RT = constant
Comparing it with ideal gas equation P =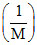 ρ RT ⇒ P = constant.
ρ RT ⇒ P = constant.
P-V graph for the cycle is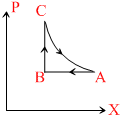 . Thus work done in cycle is positive
. Thus work done in cycle is positive
U =
Comparing it with ideal gas equation P =
P-V graph for the cycle is
 . Thus work done in cycle is positive
. Thus work done in cycle is positiveCreate a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A point source of heat of power P is placed at the center of a spherical shell of mean radius R the material of the shel...Two long, thin solid cylinders are evidential in size, but they are of different substances with two different thermal c...If S is stress and Y is Young′s modulus of material of a wire, the energy stored in the wire per unit volume is...If the adsorption coefficient and reflection of a surface of a body are 0.6 restively then : -...A Sphere of ice at 0oC having initial radius R is placed in an environ melts having ambient temperature > 0oC. The ic...