Chemical BondingHard
Question
For an exothermic chemical process occuring in two steps as
I. A + B → X (slow)
II. X → AB (fast)
the progress of the reaction can be best described by
I. A + B → X (slow)
II. X → AB (fast)
the progress of the reaction can be best described by
Options
A.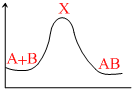

B.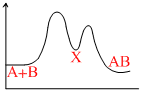

C.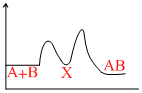

D.All are correct
More Chemical Bonding Questions
The intermolecular interaction that is dependent on the inverse cube of distance between the molecules is :-...Consider the plots, gives below, for the types of reaction:- nA → B + C (I) (II) (III)These plots respectively cor...Which of the following options represents the correct bond order ?...Consider the following sequence of reaction The product B is...Select the correct statements -...