Chemical BondingHard
Question
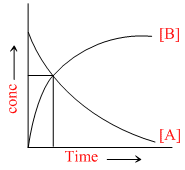
The accompanying figure depicts a change in concentration of space A and B for the reaction A → B, as a function of time. The point of inter section of the two curves represents

Options
A.t1/2
B.t3/4
C.t2/3
D.Data insufficient to predict
Solution
The intersection point indicates that half life of the reactant A is converted to B.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following compounds has the lowest melting point ?...The correct has high electron affinity ?...Which of the following options represents the correct bond order ?...The number and type of bonds between two carbon atoms in calcium carbide are...CH3CH = O + H2C = O Product (P). P is : (1 : 4)...