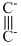Chemical BondingHard
Question
The number and type of bonds between two carbon atoms in calcium carbide are
Options
A.One sigma, one pi
B.One sigma, two pi
C.Two sigma, one pi
D.Two sigma, two pi
More Chemical Bonding Questions
Select the correct statement :...Element X is strongly electropositive and element Y is strongly electrongative. Both are univalent. The compound formed ...The correct increasing order of $C - H(A)$, $C - O(B),C = O(C)$ and $C \equiv N(D)$ bonds in terms of covalent bond leng...Match list-I with list-II and select List-I List-II(A) Zeiglernata catalyst (i) Fe4[Fe(CN)6]3(B) Brown ring complex (ii)...Which of following is not correctly matched ?...