Heat and Thermal ExpansionHard
Question
If the intermolecular force vanish away, the volume occupied by the molecules contained in 4.5 kg water at standard temperature and perssure will be given by :-
Options
A.5.6 m3
B.4.5 m3
C.11.2 litre
D.11.2 m3
Solution
If intermolecular forces vanish water behaves as gas.
Number of moles of water =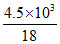 = 250
= 250
Total volume of water at STP
= 22.4 × 250 × 10-3 m3 = 5.6 m3.
Number of moles of water =
Total volume of water at STP
= 22.4 × 250 × 10-3 m3 = 5.6 m3.
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A metallic sphere having radius 0.08 m and mass m = 10 kg is heated to a temperature of 227oC and suspended inside a box...Three rods of equal length joined to from to from an equilateral triangle ABC. D is the midpoint of AB. The coefficient,...Air is filled at 60oC in s vessel of open mouth. The vessel is heat to a temperature T so that part of air escapes. The ...An ideal monotonic gas undergoes a cycle ABCA as shown in the fig. The ratio of heat absorbed during AB to the work done...Which of the following statements is /are correct ?...