Chemical Kinetics and Nuclear ChemistryHard
Question
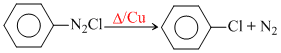
Half life is independent of the concentration of A. After 10 min volume of N2 gas is 10 L and after complete reaction is 50 L. Hence, The rate constant is
Options
A.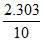 log 5 min-1
log 5 min-1
B.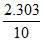 log 1.25 min-1
log 1.25 min-1
C.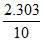 log 2 min-1
log 2 min-1
D.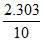 log 4 min-1
log 4 min-1
Solution
In follows first order kinetics since T50 is independent of concentration.

Hence, x = 10 L, a = 50 L
∴ k =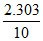 log
log 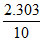 log 1.25 min-1
log 1.25 min-1

Hence, x = 10 L, a = 50 L
∴ k =
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The half-life of a radioactive isotope is three hours. If the initial mass of the isotope were 256 g, the mass of it rem...The rate of the reaction 3A + 2B → Products is given by the rate expression rate = K[A][B]2. If A is taken in large exce...The rate constant is given by the equation $K = P.A.e^{- E_{a}/RT}$. Which factor should register a decrease for the rea...Saponification of ethyl acetate (CH3COOC2H5) by NaOH (Saponification of ethyl acetate by NaOH is second order Rxn) is st...Which of the following quantity/quantities is/are temperature independent?...