Mole ConceptHard
Question
The distribution of molecular kinetic energy at two temperatures is as shown in the following graph:
Which of the following conclusions is/are correct ?
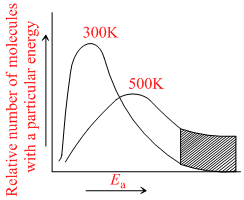
Which of the following conclusions is/are correct ?

Options
A.The number of molecules with energy Ea or greater is proportional to the shaded area for each temperature.
B.The number of molecules with energy Ea or less is proportional to the shaded area for each temperature.
C.The number of molecules with energy Ea is the mean of all temperatures.
D.The graph follows the Maxwell-Boltzmann energy distribution law.
More Mole Concept Questions
The commonly used pain reliever, aspirin, has the molecular formula C9H8O4. If a sample of aspirin contains 0.968 g of c...The density (in g mL-1) of a 3.60 M sulphuric acid solution that is 29% H2SO4 (Molar mass = 98 g mol-1) by mass will be...At 300 K, the vapour pressure of an ideal solution containing 3 mole of A and 2 mole of B is 600 torr. At the same tempe...A gaseous mixture contains 40% H2 and 60% He by volume. What is the total number of moles of gases present in 10 g of su...Law of multiple proportions is not applicable for the oxide(s) of...