Mole ConceptHard
Question
At 300 K, the vapour pressure of an ideal solution containing 3 mole of A and 2 mole of B is 600 torr. At the same temperature, if 1.5 mole of A & 0.5 mole of C (non-volatile) are added to this solution the vapour pressure of solution increases by 30 torr. Then
Options
A.PBo = 1030
B.PAo = 940
C.PAo = 1410
D.PBo = 90
Solution
PoAxA + PoBxB
600 = PoA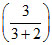 + PoB
+ PoB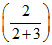 ; 3PoA + 2PoB = 3000
; 3PoA + 2PoB = 3000
& 630 = PoA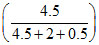 + PoB
+ PoB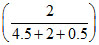
1.5 PoA = 1410 ; PoA = 940 & PoB = 90
600 = PoA
& 630 = PoA
1.5 PoA = 1410 ; PoA = 940 & PoB = 90
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
What mass of carbon disulphide CS2 can be completely oxidized to SO2 and CO2 by the oxygen liberated when 325 g of Na2O2...An organic compound "P" of molecular formula gives positive lodoform test but negative Tollen's test. When "P" is treate...Match the coloum :...A quantity of 12 g of magnesium is burnt completely in air (O2 = 20% and N2 = 80%, by volume). Which of the following is...When burnt in air, a 12.0 g mixture of carbon and sulphur yields a mixture of CO2 and SO2, in which the number of moles ...