ElectrochemistryHard
Question
Match List-I with List-II and select the correct answer using the code given below the lists.
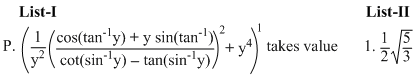
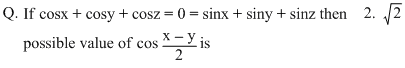
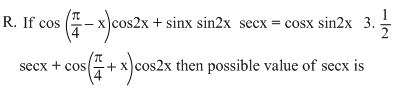
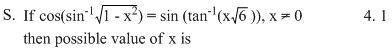
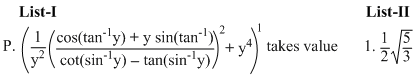
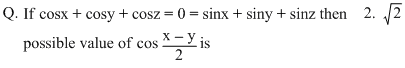
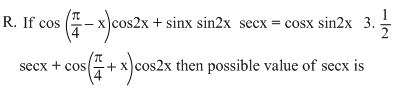
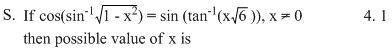
Options
A.P → 4, Q → 3, R → 1, S → 2
B.P → 4, Q → 3, R → 2, S → 1
C.P → 3, Q → 4, R → 2, S → 1
D.P → 3, Q → 4, R → 1, S → 2
Solution
(P)
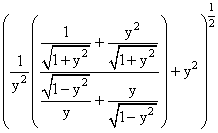
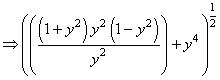
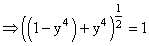
(Q)
A(cosx, sinx), B(cosy, siny) & C(cosz, sinz) lie on circle x2 + y2 = 1
∴ (0,0) is circumcentre as well as centroid of ᐃABC
⇒ ᐃABC is an equilateral triangle
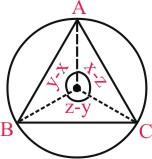
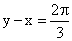
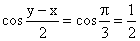
(R)
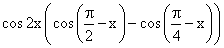 = sin 2x (1- tan x)
= sin 2x (1- tan x)
√2 sin x cos2x = sin 2x (1- tan x)
sin x(√2 cos2x - 2 (sin x - cos x)) = 0
⇒ sin x = 0 or sin x = cos x or sin x + cos x = √2
⇒ secx =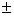 1 or sec x
1 or sec x 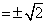
(S)
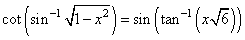
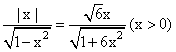
⇒ 1 - 6x2 = 6 + 6x2
⇒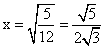
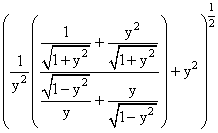
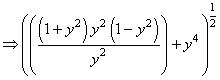
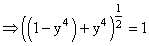
(Q)
A(cosx, sinx), B(cosy, siny) & C(cosz, sinz) lie on circle x2 + y2 = 1
∴ (0,0) is circumcentre as well as centroid of ᐃABC
⇒ ᐃABC is an equilateral triangle

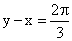
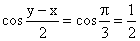
(R)
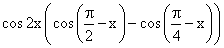 = sin 2x (1- tan x)
= sin 2x (1- tan x)√2 sin x cos2x = sin 2x (1- tan x)
sin x(√2 cos2x - 2 (sin x - cos x)) = 0
⇒ sin x = 0 or sin x = cos x or sin x + cos x = √2
⇒ secx =
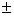 1 or sec x
1 or sec x 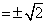
(S)
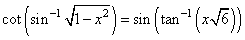
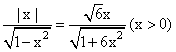
⇒ 1 - 6x2 = 6 + 6x2
⇒
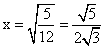
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Given : EoFe3+/Fe = - 0.036 V, EoFe2+/Fe = - 0.439V. The value of standard electrode potential for the change, Fe3+(aq) ...Choose the correct statement(s)....Pt | Cl2 (P1 atm) | HCl (0.1 M) | Cl2 (P2 atm) | Pt, cell reaction will be spontaneous if...Rusting of iron is catalyzed by...Using the standard potential values given below, decide which of the statements I, II, III, IV are correct. Choose the r...