Heat and Thermal ExpansionHard
Question
Consider the lines L1 : 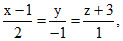 L2 :
L2 : 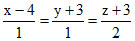 and the planes P1: 7x+y + 2z = 3, P2 : 3x + 5y - 6z = 4. Let ax + by + cz = d be the equation of the plane passing through the point of intersection of lines L1 and L2 and perpendicular to planes P1 and P2.
and the planes P1: 7x+y + 2z = 3, P2 : 3x + 5y - 6z = 4. Let ax + by + cz = d be the equation of the plane passing through the point of intersection of lines L1 and L2 and perpendicular to planes P1 and P2.
Match List-I with List-II and select the correct answer using the code given below the lists.
List-I List-II
P. a = 1. 13
Q. b = 2. -3
R. c = 3. 1
S. d = 4. -2
Match List-I with List-II and select the correct answer using the code given below the lists.
List-I List-II
P. a = 1. 13
Q. b = 2. -3
R. c = 3. 1
S. d = 4. -2
Options
A.P → 3, Q → 2, R → 4, S → 1
B.P → 1, Q → 3, R → 4, S → 2
C.P → 3, Q → 2, R → 1, S → 4
D.P → 2, Q → 4, R → 1, S → 3
Solution
For point of intersection of L1 and L2
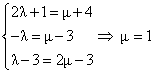 point of intersction is (5, -2, -1)
point of intersction is (5, -2, -1)
Now, vector normal to the plane is
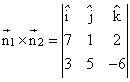
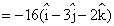
Let equation of required plane be x - 3y - 2z = α
∴ it passes through (5, -2, -1)
∴ α = 13 ⇒ equation of plane is x - 3y - 2z = 13
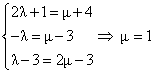 point of intersction is (5, -2, -1)
point of intersction is (5, -2, -1)Now, vector normal to the plane is
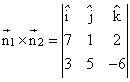
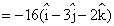
Let equation of required plane be x - 3y - 2z = α
∴ it passes through (5, -2, -1)
∴ α = 13 ⇒ equation of plane is x - 3y - 2z = 13
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A thermally insulated vessel contains some water at 0oC. The vessel connected to a vacuum pump out water vapor. This res...1023 molecules of a gas strike a target of area 1 m2 at angle 45o to normal and rebound elastically with speed 1 kms-1. ...The pressure of one mole of on mole of an ideal gas varies according to the law P = P0 - aV2, where P0 and a are positiv...Pressure versus temperature graph of an ideal gas is shown in figure. Density of the gas at point A is. ρ0 Density ...The figure shows two rods, one made of aluminum [α = 23 × 10-6 (Co)-1] and the other of steel [α = 12 ...