Heat and Thermal ExpansionHard
Question
The pressure of one mole of on mole of an ideal gas varies according to the law P = P0 - aV2, where P0 and a are positive constants. The highest temperature that the gas may may attain is :-
Options
A.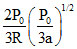
B.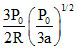
C.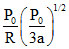
D.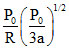
Solution
P = P0 - aV2.
From ideal gas equation
PV = nRT
⇒ RT = (P0 - aV2) V (n = 1)
⇒ T =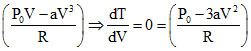
⇒ V =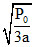 and V =
and V = 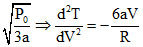 (< 0)
(< 0)
∴ Tmax =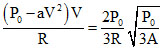
From ideal gas equation
PV = nRT
⇒ RT = (P0 - aV2) V (n = 1)
⇒ T =
⇒ V =
∴ Tmax =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
When water is boiled at 2 atm pressure, the letant heat of vaprization is 2. × 106 J/ kg and the boiling point is 1...A ring consisting of two parts ADB and ACB of same conducting k carries an amount of heat H. The ADB part is now replace...A rectangular narrow U-tube has equal arm lengths and base length, each equal to l. The verticalarms are filled with mer...Consider the MB shown in the diagram, let the resistance X have temperature coefficient and the resistance from the RB h...In the previous question, If the specific latent heat of vaporization of water at 0oC is time the specific latent heat o...