Heat and Thermal ExpansionHard
Question
The graph below shows V -P curve for three processes
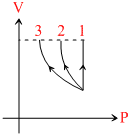
Chose the correct statement (s)

Chose the correct statement (s)
Options
A.Work done is maximum in process 1.
B.Temperature must increase in process 2 & 3.
C.Heat must be increase in process 2 & 3.
D.If final volume of gas in process 1, 2
Solution
Area under P - V curve and volume axis represent work.
Internal energy in 1 increase(expansion at constant pressure)
In process 2 and 3 internal energy may decrease depending on the process.
Since final pressure of 1, 2 & 3 ar different, final temprature must also be different.
Internal energy in 1 increase(expansion at constant pressure)
In process 2 and 3 internal energy may decrease depending on the process.
Since final pressure of 1, 2 & 3 ar different, final temprature must also be different.
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
An ideal gas expands in such a way that PV2 = constant throughout the process. Select correct alternative...Four moles of hydrogen , teo moles of helium and one mole of water vapor from an ideal gas mixture. What is the molar sp...Twelve conditioning rods from the sides of a uniform cube of side l. If in steady state, B and H ends of the cube are 10...One mole of ideal gas undergoes a cyclic process ACBA as shown in figure. Process AC is adiabatic. The temperatures at A...Water is used to cool the radiators of engines in cars because :-...