Heat and Thermal ExpansionHard
Question
One mole of ideal gas undergoes a cyclic process ACBA as shown in figure. Process AC is adiabatic. The temperatures at A, B and C are 300, 600 and 450 K respectively : - One mole of ideal gas undergoes a cyclic process ACBA as shown in figure. Process AC is adiabatic. The temperatures at A, B and C are 300, 600 and 450 K respectively :
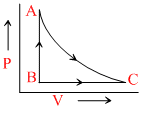

Options
A.In process CA change in internal energy is 225R.
B.In process AB change in internal energy is - 150 R.
C.In process AB change in internal energy is - 225 R.
D.Change in internal energy during the whole cyclic process is + 150 R
Solution
Ans (C)
ᐃUCA = nCV(TA - TC)
1 ×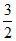 R × (300 - 450) = (- 225R)
R × (300 - 450) = (- 225R)
ᐃUAB = nCV (TB - TA)
1 ×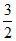 R × (600 - 300) = + 450 R
R × (600 - 300) = + 450 R
ᐃUBC = nCV (TC - TB)
1 ×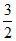 R × (450 - 600) = - 225R
R × (450 - 600) = - 225R
ᐃUABCA = ᐃUCA = ᐃUAB = ᐃUBC = 0
ᐃUCA = nCV(TA - TC)
1 ×
ᐃUAB = nCV (TB - TA)
1 ×
ᐃUBC = nCV (TC - TB)
1 ×
ᐃUABCA = ᐃUCA = ᐃUAB = ᐃUBC = 0
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Three identical rods AB, CD and PQ are joined as shown. P and Q are mid points of AB and CD respectively . Ends A, B, C ...If S is stress and Y is Young′s modulus of material of a wire, the energy stored in the wire per unit volume is...The thermo e.m.f. E in volts of a certain thermocouple is found to very with temperature difference θ in oC between...A metallic sphere having radius 0.08 m and mass m = 10 kg is heated to a temperature of 227oC and suspended inside a box...5n, n and 5n moles of a monoatomic, diatomic and non - linear polyatomic gases (which do not react chemically with each ...