ThermodynamicsHardBloom L4
Question
An ideal gas expands in such a way that PV2 = constant throughout the process. Select correct alternative
Options
A.This expansion is not possible without heating
B.This expansion is not possible without cooling
C.Internal energy remains constant in this expansion
D.Internal energy increases in the expansion
Solution
PV = nRT & PV2 = constant ⇒ V ∝ 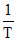 ⇒ gas can expand only it it cods
⇒ gas can expand only it it cods
As temperature decreases during expansion so internal energy will decrease will decrease.
As temperature decreases during expansion so internal energy will decrease will decrease.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
An ideal gas heat engine operates in a cornot cycle between 227oC and 127oC. It absorbs 6 kcal at the higher temperature...A gas at 50 N/m2 pressure is compressed from 10m3 to 4m3 under constant pressure. subsequently it is given 100 J of heat...If the quantity of energy with drawn from a system is δQ, work done by the system δW and the increase in inter...100g of water is heated from 30oC to 50oC. Ignoring the slight expansion of the water, the change in its internal energy...The correct equation of state for an adiabatic process is :-...