Heat and Thermal ExpansionHard
Question
The figure shows two paths for the change of state of a gas from A to B. The ratio of molar heat capacities in path 1 and path 2 is : -
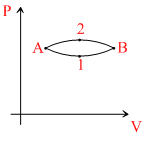

Options
A.> 1
B.< 1
C.1
D.Data insufficient
Solution
Q1 = ᐃU + W1 ; Q2 = ᐃU + W2
Ratio of specific heats
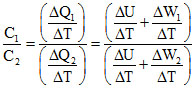 < 1 (∵W2 > W1)
< 1 (∵W2 > W1)
Ratio of specific heats
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Figure shows the adiabatic curve on log-log scale performed on a ideal gas. The gas must be :-...1023 molecules of a gas strike a target of area 1 m2 at angle 45o to normal and rebound elastically with speed 1 kms-1. ...A brass wire of length 2 m and radius 1 mm at $27^{\circ}C$ is held taut between two rigid supports. Initially it was co...A Sphere of ice at 0oC having initial radius R is placed in an environ melts having ambient temperature > 0oC. The ic...Two fine steel wires fastened between the projections of a heavy brass bar,are just taut when the whole system is at 0oC...