Heat and Thermal ExpansionHard
Question
An vessel conatins 14 g (moles) of hydrogen and 96 g (3 moles) of oxygen at ATP. Chemical reaction induced by passing electric spark in the vessel till one of the gasses is consumed. The temperature is brought back to it′s starting value k. The pressure in the vessel is -
Options
A.0.1 atm
B.0.2 atm
C.0.3 atm
D.0.4 atm
Solution
When electric spark is passed, hydrogen reacts with oxygen to from water (H2O) . Each gram of hydrogen reacts with eight grams of oxygen. Thus 96 g of oxygen will be totally consumed with 12 g hydrogen.
The gas left in the vessel will be 2g of hydrogen i.e. number of moles μ =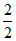 = 1.
= 1.
Using PV = μRT ⇒ P∝ μ ⇒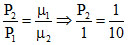 ⇒ P2 = 0.1 atm
⇒ P2 = 0.1 atm
The gas left in the vessel will be 2g of hydrogen i.e. number of moles μ =
Using PV = μRT ⇒ P∝ μ ⇒
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Two long, thin solid cylinders are evidential in size, but they are of different substances with two different thermal c...Graph a hypothetical speed distribution for a sample of N gas particle :- (for V > V0; = 0)...A sonometer wire of length 1.5 m is made of steel. The tension in it produces an elastic strain of 1%. What is the funda...A meter washer has a hole of diameter d1 and external d2, where d2 = 3d1. On heating, d2 increase by 0.3%. Then d1 will ...Some of the thermodynamic parameters are state are process variables. Some grouping of the parameter are given. Choose t...