Heat and Thermal ExpansionHard
Question
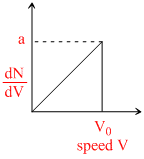
Graph a hypothetical speed distribution for a sample of N gas particle :- (for V > V0; 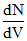 = 0)
= 0)

Options
A.The value of V0 is 2N.
B.The ration Vavg / V0 is equal to 2/3.
C.the ratio Vrms / V0 is to 1 / √2
D.Three fourth of the total particle a speed between 0.5 V0 and V0.
Solution
Hence N = 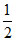 a V0 ⇒ a V0 = 2N
a V0 ⇒ a V0 = 2N
Vavg =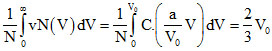
⇒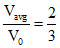
V2rms =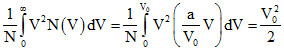
⇒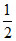
Area under the cure from 0.5 V0 to V0 is 3/4 of total area.
Vavg =
⇒
V2rms =
⇒
Area under the cure from 0.5 V0 to V0 is 3/4 of total area.
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Tree closed vessels A, Sb and C at the same temperature T and contain gasses which obey the Maxwellian distribution of v...A sonometer wire of length 1.5 m is made of steel. The tension in it produces an elastic strain of 1%. What is the funda...When two samples at different temperature are mixed, the temperature of the mixture can be : -...Two identical square rods of metal are end are welded end to end as shown in figure (a) 20 calories flows through it in ...An ideal monotonic gas undergoes a cycle ABCA as shown in the fig. The ratio of heat absorbed during AB to the work done...