Heat and Thermal ExpansionHard
Question
A refrigerator converts 100 g of water at 25oC into ice at - 10oC in one hour and 50 minutes. The quantity of heat removed per minute is (specific heat of ice = 0.5 cal /goC, latent of fusion = 80 cal /g)
Options
A.50 cal
B.100 cal
C.200 cal
D.75 cal
Solution
Heat removed in cooling water from 25oC to 0oC = 100 × 1 × 25 = 2500 cal
Heat removed in converting water into ice at 0oC = 100 × 80 = 8000 cal
Heat removed in cooling ice from 0o to -15oC = 100 × 0.5 × 10 = 500 cal
Total heat removed in 1 hr 50 min = 2500 + 8000 + 500 = 11000 cal
Heat removed per minute =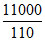 = 100 cal / min
= 100 cal / min
Heat removed in converting water into ice at 0oC = 100 × 80 = 8000 cal
Heat removed in cooling ice from 0o to -15oC = 100 × 0.5 × 10 = 500 cal
Total heat removed in 1 hr 50 min = 2500 + 8000 + 500 = 11000 cal
Heat removed per minute =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
The temperature of a body rises by 44oC when a certain of heat is given t it. The same heat when supplied to 22 g of ice...An ideal gas can be expended from an initial state to a certain volume through two different processes(i) PV2 = constant...The relation between isothermal elasticity (Eθ) and adiabatic elasticity (Eφ) is (γ = Cp/Cv) : -...Figure demonstrates a poly tropic process (i.e. PVn = constant) for an ideal gas. The work done by the gas will be in pr...Tree closed vessels A, Sb and C at the same temperature T and contain gasses which obey the Maxwellian distribution of v...