Heat and Thermal ExpansionHard
Question
An ideal gas can be expended from an initial state to a certain volume through two different processes
(i) PV2 = constant and (ii) P = KV2 where K is a positive constant. Then :-
(i) PV2 = constant and (ii) P = KV2 where K is a positive constant. Then :-
Options
A.Final temperature in (i) will be greater than in (ii)
B.Final temperature in (ii) will be greater than in (i)
C.Total heat given to the gas in case is greater than in (ii)
D.Total heat given to the gas in (i) case is greater than in (i)
Solution
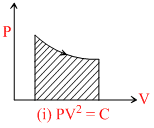
(i) PV2 = C ⇒ TV = C
If volume expands temperature decrease.
(ii) P = KV2 ⇒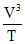 = constant
= constant
If volume expands, temprature increase
Q = ᐃU + W
Q2 > Q as W2 W2 > ᐃU2 > ᐃU1
If volume expands temperature decrease.
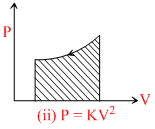
(ii) P = KV2 ⇒
If volume expands, temprature increase


Q = ᐃU + W
Q2 > Q as W2 W2 > ᐃU2 > ᐃU1
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Three long straight wires carrying current are arranged mutually parallel as shown in the figure. The force experienced ...The figure a system of two constrict spheres of radii r1 and r2 and kept at temperatures T1 and T2, respectively. the ra...The variation of lengths of two metal rods A and B with change in temperature is shown in figure. The ratio of is : -...The pressure that has to be applied to the ends of a steel rod of length 10 cm to keep its length constant when its temp...At temperature T, N molecules of gas A each having mass m and and at the same temperature 2N molecules of gas B each hav...