Solid StateHard
Question
The radius ratio of P2+ and Q- ions 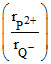 is 0.8. The unit cell of this ionic solid is
is 0.8. The unit cell of this ionic solid is
Options
A.Simple cubic for Q- ions and P2+ ions occupy all the cubic voids
B.Face centered cubic for Q- ions and P2+ ions occupy all the tetrahedral voids.
C.Face centered cubic for P2+ ions and Q- ions occupy all the tetrahedral voids.
D.Face centered cubic for Q- ions and P2+ ions occupy 50% of octahedral voids.
More Solid State Questions
A mineral having the formula AB2 crystallizes in the ccp lattice, with A atoms occupying the lattice points. Select the ...Which of the following is/are correct ?...The unit cell cube length for LiCI (just like NaCI structure) is 5.14 A0. Assuming anion–anion contact, the ionic radius...In a sodium chloride crystal, the unit cell edge length is a. The option(s) representing the correct combination is/areO...Which of the following is/are pseudo solids ?I. KCI II. Barium chloride dihydrateIII. RubberIV. Solid cake left after di...