Solid StateHard
Question
The spinal structure (AB2O4) consists of an fcc array of O2- ions in which the :
Options
A.A cation occupies one-eighth of the tetrahedral holes and B cation occupies one-half of octahedral holes
B.A cation occupies one-fourth of the tetrahedral holes and the B cations the octahedral holes
C.A cation occupies one-eighth of the octahedral hole and the B cation the tetrahedral holes
D.A cation occupies one-fourth of the octahedral holes and the B cations the tetrahedral holes
Solution
A → 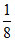 × 8 = 1, B → 4 ×
× 8 = 1, B → 4 × 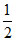 = 2 and O2- = 4 so formula of spinal = AB2O4
= 2 and O2- = 4 so formula of spinal = AB2O4
Create a free account to view solution
View Solution FreeMore Solid State Questions
In the zinc blend structure (ZnS), S2− adopts CCP arrangement and Zn2+ occupies...An element (atomic mass = 100 g/mole) having BCC structure has unit cell edge 400 pm. The density of the element is (no....Percentage of free space in cubic close packed structure and in body centred packed structure are respectively...In a sodium chloride structure, the percentage of the octahedral voids occupied by cations is :...The composition of a sample of cuprous oxide is found to be Cu1.92O1.00 due to metal deficient defect. The molar ratio o...