Solid StateHard
Question
In a hypothetical solid, C atoms are found to form cubical close-packed lattice. A atoms occupy all tetrahedral voids and B atoms occupy all octahedral voids.
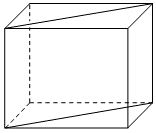
A and B atoms are of appropriate size, so that there is no distortion in the ccp lattice of C atoms. Now if a plane as shown in the following figure is cut, then the cross section of this plane will look like

A and B atoms are of appropriate size, so that there is no distortion in the ccp lattice of C atoms. Now if a plane as shown in the following figure is cut, then the cross section of this plane will look like
Options
A.

B.

C.

D.

Solution
From figure it is clear 4 corners and 2 face centers lie on the shaded plane. Therefore, there will be six C atoms, and atoms (marked A) in TVs do not touch other.
Fig. (A) is not possible; four atom marked C.
Fig. (B) is not possible, atoms A in TVs are not shown in figure.
Fig. (C) is possible, since atoms A in TVs are not touching each other.
Fig. (D) is not possible, since atoms A in TVs are touching each other.
Fig. (A) is not possible; four atom marked C.
Fig. (B) is not possible, atoms A in TVs are not shown in figure.
Fig. (C) is possible, since atoms A in TVs are not touching each other.
Fig. (D) is not possible, since atoms A in TVs are touching each other.
Create a free account to view solution
View Solution FreeMore Solid State Questions
Which of the following statements about the interstitial compounds is incorrect ?...Which of the following compounds containing carbonyl group will give coloured crystalline product with...An ionic solid is HCP of Q2− ions and Px+ ions are in half of the tetrahedral voids. The value of x should be...Three lines are drawn from a single corner of an FCC unit cell to meet the other corner such that they are found to pass...Following three planes (P1, P2, P3) in an fc unit cell are shown in the figure below. Consider the following statements ...