Solid StateHard
Question
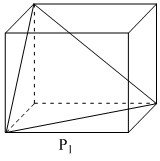
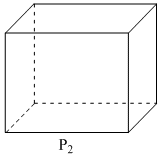
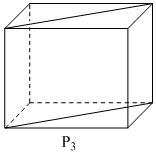
Following three planes (P1, P2, P3) in an fc unit cell are shown in the figure below. Consider the following statements and choose the correct option/options that follow:



Options
A.P1 contains no three dimensional voids.
B.P2 contains only octahedral voids.
C.P3 contains both octahedral and tetrahedral voids.
D.All of these
Solution
A. P1 represent one of the close-packed layer having triangular voids only.
B. P2 contains location of OVs (edge centers of unit cell).
C. P3 contains 3 OVs locations (one at body center and two at edge center). Also, plane
P3 contains the body diagonals,hence it contains TVs location(TVs lie at body diagonal)
B. P2 contains location of OVs (edge centers of unit cell).
C. P3 contains 3 OVs locations (one at body center and two at edge center). Also, plane
P3 contains the body diagonals,hence it contains TVs location(TVs lie at body diagonal)
Create a free account to view solution
View Solution FreeMore Solid State Questions
Which of the following statement(s) for crystal having schottky defect is/are correct....If the height of HCP unit cell of identical particles is h, then the height of octahedral voids from the base is...If the anions (A) form hexagonal closest packing and cations (C) occupy only 2/3 octahedral voids in it, then the genera...A metal crystallizes in such a lattice in which only 70% of the total space of the crystal is occupied by the atoms. If ......