SolutionHard
Question
64.4g Crystals of pure Na2SO4 10 H2O are dissolved in 114 g of water to obtain an aq. solution of salt. Determine the freezing point of solution assuming that salt is completely ionized in solution Kf of water is 1.86 K kgmol-1 :-
Options
A.-7.44oC
B.-9.79oC
C.-3.26oC
D.-2.48oC
Solution
Moles of solute (n) = 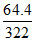 = 0.2 mol Na2SO4 10 H2O
= 0.2 mol Na2SO4 10 H2O
∵ 1 mol Na2SO4 10H2O contains = 10 mol H2O
⇒ 180 gH2O
∴ 0.2 mol Na2SO4 10H2O contains = 180 × 0.2 = 36 g H2O
Total mass of solvent (H2O) in solu. = 114 + 36 = 150 g
∴ ᐃTf = 3 × 1.86 ×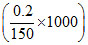 = 7.44
= 7.44
∵ 1 mol Na2SO4 10H2O contains = 10 mol H2O
⇒ 180 gH2O
∴ 0.2 mol Na2SO4 10H2O contains = 180 × 0.2 = 36 g H2O
Total mass of solvent (H2O) in solu. = 114 + 36 = 150 g
∴ ᐃTf = 3 × 1.86 ×
Create a free account to view solution
View Solution FreeMore Solution Questions
The melting points of most of the solid substances increases with an increase of pressure acting on them. However, ice m...Which of the following mixture of solutions has pH = 1.0 ?...Which of the following shows negative deviation from Raoult′s law ?...An aqueous solution of a solute AB has b.p. of 101.08oC (AB is 100% ionised at boiling point of the solution) and freeze...Water should boil at 90°C at pressure...