SolutionHard
Question
Which of the following mixture of solutions has pH = 1.0 ?
Options
A.100 mL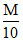 HCl + 100 mL
HCl + 100 mL 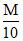 NaOH
NaOH
B.55 mL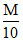 HCl + 45 mL
HCl + 45 mL 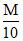 NaOH
NaOH
C.10 mL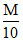 HCl + 90 mL
HCl + 90 mL 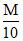 NaOH
NaOH
D.75 mL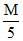 HCl + 25 mL
HCl + 25 mL 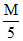 NaOH
NaOH
Solution
HCl + NaOH → NaCl + H2O
M1V1 M2V2
(A) 10 10 Complete neutralization
pH = 7
(B) 5.5 4.5 [H+] =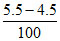
pH = 2
(C) 1 9 [OH-] =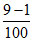
pH = 12.9
(D) 15 5 [H+] =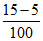
pH = 1
M1V1 M2V2
(A) 10 10 Complete neutralization
pH = 7
(B) 5.5 4.5 [H+] =
pH = 2
(C) 1 9 [OH-] =
pH = 12.9
(D) 15 5 [H+] =
pH = 1
Create a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressures of pure liquids A, B and C are 75, 22 and 10 torr, respectively. Which of the following is/are poss...A solution of x moles of sucrose in 100 grams of water freezes at -0.2oC. As ice separates the freezing point goes down ...The vapour pressure of water depends upon :...Benzene and toluene form an ideal solution. The vapour pressures of benzene and toluene are 75 mm and 25 mm, respectivel...Water should boil at 90°C at pressure...