Atomic StructureHard
Question
The energies E1 and E2 of tworadiations are 25 eV and 50 eV respectively. The relation between their wavelengths i.e., λ1 and λ2 will be
Options
A.λ1 = 2λ2
B.λ1 = 4λ2
C.λ1 = 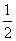 λ2
λ2
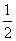 λ2
λ2D.λ1 = λ2
Solution
E1 = 25 eV, E2 = 50 eV
E1 =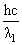 and E2 =
and E2 = 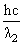
or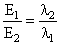
or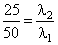
or λ1 = 2λ2
E1 =
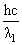 and E2 =
and E2 = 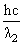
or
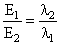
or
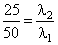
or λ1 = 2λ2
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The potential energy of electron revolving in the ground state of H atom is...The K.E. of electron in He+ will be maximum in...In different experiments, α-particles, proton, deuteron and neutron are projected towards gold nucleus with the same ki...A lithium atom has three electrons. Assume the following simple picture of the atom. Two electrons move close to the nuc...An electron at rest is accelerated through a potential difference of 200 V. If the specific charge of electron is 1.764...