Chemical EquilibriumHard
Question
For the reaction N2(g) + O2(g) ⇋ 2NO (g), the equilibrium constant is K1. The equilibrium constant is K2 for the reaction 2NO (g) + O2(g) ⇋ 2NO2(g), What is K for the reaction NO2(g) ⇋ 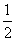 N2(g) + O2(g) ?
N2(g) + O2(g) ?
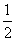 N2(g) + O2(g) ?
N2(g) + O2(g) ?Options
A.1/( 4 K1 K2 )
B.[1/( K1 K2)]1/2
C.1/( K1 K2 )
D.1/( 2 K1 K2 )
Solution
N2(g )+ O2(g) ⇋ 2NO (g); K1 ....(i)
2NO (g) + O2(g) ⇋ 2NO2(g); K2 ....(i)

N2(g) + O2(g) ⇋ 2NO2(g); K = K1 × K2
∴ For NO2(g) ⇋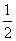 N2(g) + O2(g); K =
N2(g) + O2(g); K = 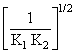
2NO (g) + O2(g) ⇋ 2NO2(g); K2 ....(i)

N2(g) + O2(g) ⇋ 2NO2(g); K = K1 × K2
∴ For NO2(g) ⇋
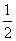 N2(g) + O2(g); K =
N2(g) + O2(g); K = 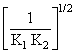
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
What will be the enthalpy change for the reaction (in kcal/mol)2H2(g) + O2(g) + 2H2O(l) → 4H+(aq) + 4OH- (aq.)Give...What is the equilibrium expression for the reaction P4(s) + 5O2(g) ⇋ P4O10(s) ?...A gaseous mixture contains 0.30 moles of CO, 0.10 moles of H2, and 0.03 moles of H2O vapour and an unknown amount of CH4...What can you say about the existance of A if the potential energy diagram for the reaction A → B looks like...The value of KP for the process: CuSO4·5H2O(s) $\rightleftharpoons$CuSO4·3H2O(s) + 2H2O(g) is 1.21 × 10–4 atm2 at certai...