Gaseous StateHard
Question
Two gases A and B having the same volume diffuse through a porous partition in 20 and 10 seconds respectively. He molecuar mass of A is 46 u. Molecular mass of B will be
Options
A.12.25 u
B.6.50 u
C.25.00 u
D.50.00 u
More Gaseous State Questions
The Ca2+ and F- are located in Caf crystal, respectively at face centred cubic lattice points and in...Calculate the entropy change involved in the isothermal reversible expansion of 5 mol of ideal gas from a volume of 10L ...A 40 ml of a mixture of H2 and O2 at 18oC and 1 atm pressure was sparked so that the formation of water was complete. Th...Match of following (where Urms = root mean square speed, Uav = average speed, Ump = most probable speed) List I List II ...Which of the following combination will produce H2 gas?...
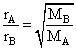
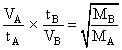
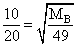
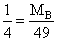
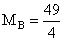 = 12.25 u.
= 12.25 u.