ThermodynamicsHard
Question
For the cell, Mn(s)|Mn(aq)+2(0.4M)||Sn(aq)+2(0.04)|Sn(s), Calculate free energy change (ᐃG) at 298 K. Given : EoMn+2|Mn = - 1.18 V; EoSn+2|Sn = - 0.14 volt 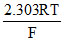 = 0.06
= 0.06
Options
A.180.93 KJ
B.- 194.93 KJ
C.-180.93 KJ
D.None of these
More Thermodynamics Questions
For a particular reversible reaction at temperature T, ᐃH and ᐃS were found to be both +ve. If Te is the tem...Select the correct statements....If liquids A and B form an ideal solution...Which of the following statement(s) is/are false ?...The value of log10K for a reaction A ⇋ B is ( Given : áƒfHo298K = - 54.07 kJ mol-1, áƒrSo298K = 10JK-1mol-1 and R = ...