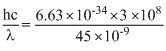AIPMT | 2014Atomic StructureHard
Question
Calculate the energy in joule corresponding to light of wavelength 45 nm : (Planck′s constant h = 6.63 × 10-34 Js; speed of light c = 3 × 108 ms-1)
Options
A.6.67 × 10ms15
B.6.67 × 10ms11
C.4.42 × 10ms-15
D.4.42 × 10ms-18
More Atomic Structure Questions
Which of the following will have magnetic moment about 4.9 BM?...M-2 configuration is 1s2 2s2 2p6 then M+2 configuration will be :-...Some hydrogen-like atoms in ground state absorbs ‘n’ photons having same the energy and on de-excitement, it emits exact...Maximum number of electrons in a subshell with l = 3 and n = 4 is:...The frequency of certain line of the Lyman series (n2 = 4 to n1 = 1) of the atomic spectrum of hydrogen can satisfy the ...