Atomic StructureHard
Question
The I.P. of an atom is 16 eV. What is the value of first excitation potential :
Options
A.10.2 eV
B.12 eV
C.14 eV
D.16 eV
Solution
E∞ - E1 = 16 eV ⇒ E1 = - 16eV
E2 =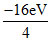 = - 4eV
= - 4eV
Ist excitation potential = E2 - E1
= - 4eV - (-16eV)
= 12 eV
E2 =
Ist excitation potential = E2 - E1
= - 4eV - (-16eV)
= 12 eV
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Electrons are de-exciting from the fifth orbit in hydrogen atoms but the first orbit is not available for them. The maxi...The angular momentum of electron in Bohr′s orbit is J. What will be the potential energy of that Bohr′s orbi...Which of the following is not a correct statement according to Rutherford’s atomic model?...Which is/are correct for sodium atom in the ground state?...If e = 1.60206 × 10-19 C, = 1.75875 × 1011C kg-1then mass of electron is...