Atomic StructureHard
Question
The angular momentum of electron in Bohr′s orbit is J. What will be the potential energy of that Bohr′s orbit :-
Options
A.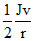
B.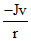
C.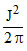
D.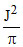
Solution
⇒ we know PE = - 2 × K.E.
Now J = m v r
or Jv = m v2 r
or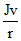 mv2 = 2 ×
mv2 = 2 × 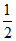 mv2 = 2 × KE = - PE
mv2 = 2 × KE = - PE
so, PE = -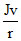
Now J = m v r
or Jv = m v2 r
or
so, PE = -
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A hydrogen - like atom has ground state binding energy 122.4 eV. Then :...The group in which all the elements do not have same number of electrons in their valence shell is :-...After np orbitals are filled, the next orbital filled will be :...The orbital angular momentum of an electron in 2s-orbital is :...Quantum numbers of an atom can be defined on the basis of...