Ionic EquilibriumHard
Question
Which of the following weakest base ?
Options
A.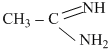
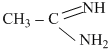
B.CH3 - CH = NH
C.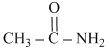
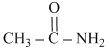
D.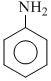

More Ionic Equilibrium Questions
50 mL of 0.1 M NaOH is added to 60 mL of 0.15 M H3PO4 solution (K1, K2 and K3 for H3PO4 are 10-3, 10-8 and 10-13 respect...Which of the following solution will have a pH exactly equal to 8 ?...Most reactive towards Nucleophilic attack -...A solution containing a mixture of 0.05 M NaCl and 0.05 M NaI is taken (Ksp of AgCl = 10-10 and Ksp of AgI = 4 × 10...The dissociation constant of formic acid is 0.00024. The hydrogen ion concentration in 0.002 M-HCOOH solution is nearly...