Boron and Carbon FamilyHard
Question
Calculate the partial pressure of carbon monoxide from the following :-
CaCO3(s) ⇋ CaO(s) + CO2 (g) ; KP = 8 × 10-2
CO2(g) + C(s) ⇋ 2CO(g) ; KP = 2
CaCO3(s) ⇋ CaO(s) + CO2 (g) ; KP = 8 × 10-2
CO2(g) + C(s) ⇋ 2CO(g) ; KP = 2
Options
A.0.2
B.0.4
C.1.6
D.4
Solution
From equation (i) Kp = PCO2 ⇒ PCO2 = 8 × 10-2
from equation (ii) Kp =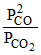
⇒ PCO =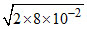 = 4 × 10-1 = 0.4 atm
= 4 × 10-1 = 0.4 atm
from equation (ii) Kp =
⇒ PCO =
Create a free account to view solution
View Solution FreeMore Boron and Carbon Family Questions
What volume of hydrogen gas, at 273 K and 1 atm, pressure will be consumed in obtaining 21.6 g of elemental boron (atomi...Heating an aqueous solution of aluminium chloride to dryness will give...Thermodynamically the most stable form of carbon is...On heating to 400-500oC relatively unstable hydrides and carbonates decompose. Which of the following will decompose whe...Proceeding from the enthalpy of formation of gaseous CO2(ᐃHfo = - 393.5 kJ mol-1) and the thermochemical equation ...