Boron and Carbon FamilyHard
Question
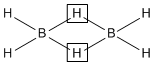
The structure of diborane (B2H6) contains
Options
A.four 2c-2e bonds and two 3c-2e bonds
B.two 2c-2e bonds and four 3c-2e bonds
C.two 2c-2e bonds and two 3c-3e bonds
D.four 2c-2e bonds and four 3c-2e bonds
More Boron and Carbon Family Questions
In the structure of diamond, carbon atoms appears at...Cross link in silicone can be induced with :-...From the following, the least stable structure is :...Which is non linear :-...A dipeptide is obtained by Glycine & Alanine.wherever it is treated with in presence of sodium carbonate, it gives follo...