Solid StateHard
Question
Following three planes (P1, P2, P3) in an FCC unit cell are shown. Consider the following statements and choose the correct option that follow :
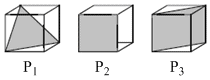

Options
A.P1 contains triangular void only
B.P2 contains only octahedral voids
C.P3 contains both octahedral and tetrahedral voids
D.None of these
More Solid State Questions
An element crystallises in a ′bcc′ lattice. Nearest neighbours and next nearest neighbours of the elements a...The correct statement(s) for the packing of identical spheres in two dimensions is/ are...CsCl crystallises in body centred cubic lattice. If ′a′ is its edge length then which of the following expre...A solid contains An+ and Bm− ions. The structure of solid is FCC for Bm− ions and An+ ions are present in one-fourth of ...How many units cells are there in 1.00 g cube shaped ideal crystal of AB (Mw = 60) which has a NaCl type lattice...