JEE Main | 2014Solid StateHard
Question
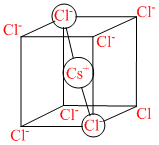
CsCl crystallises in body centred cubic lattice. If ′a′ is its edge length then which of the following expressions is correct?
Options
A.rCs+ + rCl- = 3a
B.rCs+ + rCl- = 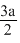
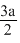
C.rCs+ + rCl- = 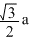
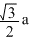
D.rCs+ + rCl- = √3a
More Solid State Questions
MgO exists in a rock salt type unit cell. Each Mg2+ ion will be in contact with...Which kind of defects is introduced by doping in silicon crystal?...The number of octahedral void(s) per atom present in a cubic close-packed structure is...In the calcium fluoride structure, the coordination numbers of calcium and fluoride ions are, respectively,...In an FCC unit cell a cube is formed by joining the centers of all the tetrahedral voids to generate a new cube.Then the...