ThermodynamicsHard
Question
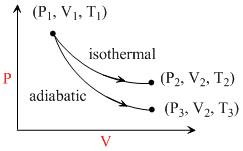
The reversible expansion of an ideal gas under adiabatic and isothermal conditions is shown in the figure.Which of the following statement(s) is (are) correct?

Options
A.T3 > T1
B.T1 = T2
C.wisothermal > wadiabatic
D.ᐃUisothermal > ᐃUadiabatic
Solution
T1 = T2 because process is isothermal.
Work done in adiabatic process is less than in isothermal process because area covered by isothermal curve is more than the area covered by the adiabatic curve.
In adiabatic process expansion occurs by using internal energy hence it decreases while in isothermal process temperature remains constant that′s why no change in internal energy.
Work done in adiabatic process is less than in isothermal process because area covered by isothermal curve is more than the area covered by the adiabatic curve.
In adiabatic process expansion occurs by using internal energy hence it decreases while in isothermal process temperature remains constant that′s why no change in internal energy.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A sample of ideal gas is compressed from initial volume of 2Vo to Vo using three different processes as follows. (1) Rev...Which of the following sets does show the intensive properties?...Which of the following is correct option for free expansion of an ideal gas under adiabatic condition ?...An ideal gas in a thermally insulated vessel at internal pressure = P1, volume = V1 and absolute temperature = T1 expand...If all degree of freedom of a three- dimensional N-atomic gaseous molecules is excited, then CP/CV ratio of gas should b...