JEE Advanced | 2014ThermodynamicsHard
Question
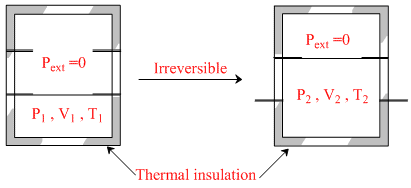
An ideal gas in a thermally insulated vessel at internal pressure = P1, volume = V1 and absolute temperature = T1 expands irreversibly against zero external pressure, as shown in the diagram. The final internal pressure, volume and absolute temperature of the gas are P2, V2 and T2, respectively. For this expansion,

Options
A.q = 0
B.T2 = T1
C.P2V2 = P1V1
D.P2V2γ = P1V1γ
Solution
Work against zero external pressure is zero and q = 0 due to insulated boundary
So, q = 0
ᐃU = 0
ᐃT = 0
∴T2 = T1
and P2V2 = P1V1
So, q = 0
ᐃU = 0
ᐃT = 0
∴T2 = T1
and P2V2 = P1V1
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
An ideal monoatomic gas initially at 300 K expands adiabatically into vacuum to double its volume. The final temperature...A system absorbs 20 kJ heat and does 10 kJ of work. The internal energy of the system...The direct conversion of A to B is difficult, hence it is carried out by the following shown path: Given ᐃS(A U...Which of the following sets does show the intensive properties?...Molar heat capacity of CD2O (deuterated form of formaldehyde) vapour at constant pressure is vapour 14 cal/K-mol. The en...