Chemical Kinetics and Nuclear ChemistryHard
Question
In the reaction N2 + 3H2 ⇋ 2NH3
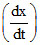 = 1 × 102 [N2] [H2]3 - 1 × 10-3 [NH3]2 and at some instant if
= 1 × 102 [N2] [H2]3 - 1 × 10-3 [NH3]2 and at some instant if 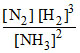 = 10-5 M2
= 10-5 M2
then at this instant value of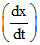 is :
is :
then at this instant value of
Options
A.0
B.1 × 105
C.1 × 10-5
D.1 × 10-3
Solution
Given Rxn
N2 + 3 H2 ⇋ 2 NH3
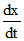 = 1 × 102 [N2] [H2]3 - 1 × 10-3 [NH3]2
= 1 × 102 [N2] [H2]3 - 1 × 10-3 [NH3]2
when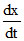 = 0 then
= 0 then 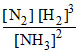 = 10-5
= 10-5
Hence the reaction will be at equilibrium at the given instant.
N2 + 3 H2 ⇋ 2 NH3
when
Hence the reaction will be at equilibrium at the given instant.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Two reaction, A → products and B → products have rate constants Ka and Kb at temperature T and activation en...According to the collisions theory, the rate of reaction increases with temperature due to...In the above question solution is optically inactive when :...The reaction H3C−CH2−NO2 + OH− → H3C− CH− −NO2 + H2O obeys the rate law for pseudo first-order kinetics in the presence ...Milk is colloid in which :...