ThermodynamicsHard
Question
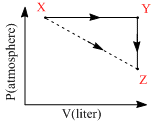
For an ideal gas, consider only P-V work in going from an initial state X to the final state Z. The final stateZ can be reached by either of the two paths shown in the figure. Which of the following choice(s) is(are)correct? [Take ᐃS as change in entropy and w as work done]

Options
A.ᐃSx→z = ᐃSx→y + ᐃSy→z
B.Wx→z = Wx→y + Wy→z
C.Wx→y→z = Wx→y
D.ᐃSx→y→z = ᐃSx→y
Solution
ᐃSx→z = ᐃSx→y + ᐃSY→Z [entropy S is a state function, hence additive]
WX→Y→Z = WX→Y (work done in Y → Z is zero as it is an isochoric process)
WX→Y→Z = WX→Y (work done in Y → Z is zero as it is an isochoric process)
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Which of the following processes takes place with decrease of entropy ?...Variation in gene frequencies within populations can occur by chance rather than by natural selection.This is referred t...Among the following, the intensive property is (properties are)...Standard enthalpy of vaporisation ᐃvapHo for water at 100oC is 40.66 kJ mol-1. The internal energy of vaporisation...The direct conversion of A to B is difficult, hence it is carried out by the following shown path: Given ᐃS(A U...