ThermodynamicsHard
Question
Standard enthalpy of vaporisation ᐃvapHo for water at 100oC is 40.66 kJ mol-1. The internal energy of vaporisation of water at 100oC (in kJ mol-1) is
(Assume water vapour to behave like an ideal gas).
(Assume water vapour to behave like an ideal gas).
Options
A.+ 37.56
B.- 43.76
C.+ 43.76
D.- 40.66
Solution
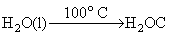
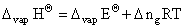
For the above reqaction,
ᐃ ng = np - nr = 1 - 0 = 1
∴ 40.66 kJ mol-1 =
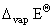 + 1 × 8.314 × 10-3 × 373
+ 1 × 8.314 × 10-3 × 373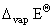 = 40.66 kJ mol-1 - 3.1 kJ mol-1
= 40.66 kJ mol-1 - 3.1 kJ mol-1= + 37.56 kJ mol-1
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The entropy change in the fusion of one mole of a solid melting at 300 K (latent heat of fusion, 2930 J/mol) is...Assume ideal gas behaviour for all the gases considered and vibrational degrees of freedom to be active. Separate equimo...The change in entropy accompanying the heating of one mole of helium gas (Cv,m = 3R/2), assumed ideal, from a temperatur...One mole of an ideal monoatomic gas is heated in a process PV5/2 = constant. By what amount heat is absorbed in the proc...When the value of entropy is greater, then the ability for work is...