Atomic StructureHard
Question
Match the following
(a) Energy of ground state of He+ (i) + 6.04 eV
(b) Potential energy of I orbit of H-atom (ii) -27.2 eV
(c) Kinetic energy of II excited state of He+ (iii) 54.4 V
(d) Ionisation potential of He+ (iv) - 54.4 eV
(a) Energy of ground state of He+ (i) + 6.04 eV
(b) Potential energy of I orbit of H-atom (ii) -27.2 eV
(c) Kinetic energy of II excited state of He+ (iii) 54.4 V
(d) Ionisation potential of He+ (iv) - 54.4 eV
Options
A.A - (i), B - (ii), C - (iii), D - (iv)
B.A - (iv), B - (iii), C - (ii), D - (i)
C.A - (iv), B - (ii), C - (i), D - (iii)
D.A - (ii), B - (iii), C - (i), D - (iv)
Solution
(a) Energy of ground state of He+ = - 13.6 × 22 = - 54.4 eV (iv)
(b) Potential energy of I orbit of H-atom = - 27.2 × 12 = - 27.2 eV (ii)
(c) Kinetic energy of II excited state of He+ = 13.6 ×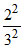 = 6.04 eV (i)
= 6.04 eV (i)
(d) Ionisation potential of He+ = 13.6 × 22 = 54.4 V (iii)
(b) Potential energy of I orbit of H-atom = - 27.2 × 12 = - 27.2 eV (ii)
(c) Kinetic energy of II excited state of He+ = 13.6 ×
(d) Ionisation potential of He+ = 13.6 × 22 = 54.4 V (iii)
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
How many of the following compounds have used their d-orbital in hybridisationCF4, SF4, I3-, SOF4, PF3Cl2, SnCl2, SO42-...The number of d-electrons in Fe2+ (Z = 26) is not equal to the number of electrons in which one of the following?...A photon of 300 nm is absorbed by a gas and then emits two photons. One photon has a wavelength 496 nm then the waveleng...The principal quantum number of an atom is related to the :...If the radius of second Bohr orbit is x, then de Broglie wavelength of electron in 3rd orbit is nearly :...