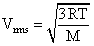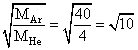KTGHard
Question
A mixture of 2 moles of helium gas (atomic mass = 4 amu) and 1 mole of argon gas (atomic mass = 40amu) is kept at 300 K in a container. The ratio of the rms speeds 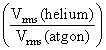 is
is
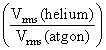 is
isOptions
A.0.32
B.0.45
C.2.24
D.3.16
More KTG Questions
A certain mass of an ideal gas is at pressure P1 and volume V1. It is compressed isothermally and then allowed to expand...For a real gas, the following is true -...At 27o C temperature, the kinetic energy of an ideal gas is E1. If the temperature is increased to 327o C, then kinetic ...Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will...Air is filled at 60o C in a vessel of open mouth. The vessel in heated to a temperature T so that 1/4th part of air esca...