KTGHard
Question
A certain mass of an ideal gas is at pressure P1 and volume V1. It is compressed isothermally and then allowed to expand adiabatically until its pressure returns to P1. The gas is then allowed to expand its original volume. Which of the following P−V graphs are these process correctly shown?
Options
A.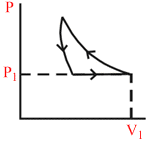

B.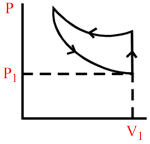

C.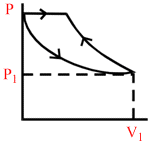

D.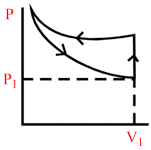

More KTG Questions
At 27o C temperature, the kinetic energy of an ideal gas is E1. If the temperature is increased to 327o C, then kinetic ...A gas behaves more closely like an ideal gas at...The average translational kinetic energy of molecule depends on :-...If the ratio of specific heats is γ and R is the gas constant then what will be the value of specific heat for gas ...Which of the follwing will be same if temperature of two ideal gas is equal...