Carboxylic Acid and Acid DerivativesHard
Question
Which of the following statement is not true ?
Options
A.At room temperature, formal chloride is present in the of CO and HCl
B.Acetamide behaves as a weak base as well as a weak acid.
C.CH3CONH2  CH3CH2NH2
CH3CH2NH2
 CH3CH2NH2
CH3CH2NH2D.All of the three
Solution
(a) 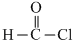 → CO + HCl
→ CO + HCl
(b) CH3 CONH2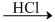 CH3CON+H3Cl-
CH3CON+H3Cl-
(Acetamide as a weak base)
(c) CH3CPNH2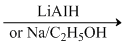 CH3CH2NH2 + H2O
CH3CH2NH2 + H2O
Thus all the three are correct.
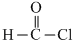 → CO + HCl
→ CO + HCl(b) CH3 CONH2
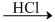 CH3CON+H3Cl-
CH3CON+H3Cl-(Acetamide as a weak base)
(c) CH3CPNH2
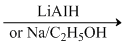 CH3CH2NH2 + H2O
CH3CH2NH2 + H2OThus all the three are correct.
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
CH3COCl + C2H5O CH3COOC2H5 + HCl. The function of pyridine in the above reaction is -...Let us consider an esterification of isotopically labelled carboxylic acid - + CH3CH2OH (x) and (Y), (X) and (Y) respect...Esterification is fastest in case of -...Which of the following is the best representation of the structure of the carboxylate ion -...3g of activated charcoal was added to 50 mL of acetic acid solution (0.06N) in a flask. After an hour it was filtered an...